Before answering this question it helps to first know how much a gallon of gasoline weighs.
Gasoline has a weight of just over 6 pounds per US gallon. A natural guess could then be that about 6 pounds of carbon dioxide (CO2) is produced from the combustion of a gallon of gasoline. However, to the surprise of many, that guess would actually be too low.
Gasoline is comprised primarily of carbon and hydrogen atoms linked together forming large carbon chain molecules. During the combustion process, the chemical bonds linking individual carbon atoms together are broken apart, releasing energy that is used to power the vehicle’s engine. When the bond between two carbon atoms is broken, each of those freed carbon atoms then bond naturally with other atoms in the atmosphere, typically oxygen atoms. From this chemical reaction we find the answer to our question.
A single carbon atom has a molecular weight of 12. After combustion, the vast majority of carbon atoms in a gallon of gasoline bond with two oxygen atoms, forming carbon dioxide (CO2) molecules. An oxygen atom has a molecular weight of 16. Therefore, a carbon dioxide molecule has a molecular weight of 44. So a single carbon dioxide molecule is 3.6 times heavier than a single carbon atom.
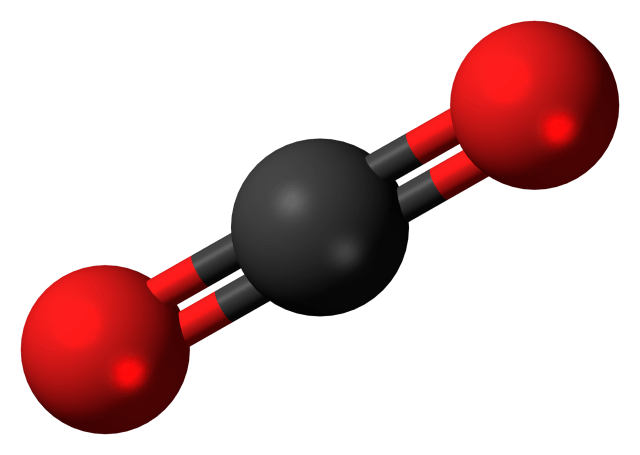
Carbon dioxide molecule comprised of two oxygen atoms (red) and one carbon atom (black). Image Credit: https://upload.wikimedia.org/wikipedia/commons/ a/a0/Carbon_dioxide_3D_ball.png
The EPA cites that the carbon content of a gallon of gasoline is 5.3 pounds (2,400 grams). If each carbon atom in a gallon of gasoline becomes 3.6 times heavier when converted to carbon dioxide after combustion, then the 5.3 pounds of carbon in a gallon of gas becomes just over 19 pounds (8,650 grams) of carbon dioxide.
So the answer to the question posed at the beginning of this post is that after combustion, one gallon of gasoline weighing about 6 pounds, forms just over 19 pounds of carbon dioxide.
Put in perspective, when an 18 gallon tank is filled at the pump, that car leaves the gas station about 108 pounds heavier. Once that tank of gasoline has been consumed, that 100 pounds of gasoline has been converted into over 340 pounds of carbon dioxide and deposited into the atmosphere.

Pingback: Fixing the atmosphere: can a common rock help? | New York
Reblogged this on Energy post.
thank you for great post
Just curious, is it possible three years later to correct mistake in co2 molecule. Wording says two carbon molecules plus one carbon molecule.
Thanks for noticing the typo. We have fixed this.
Pingback: Questions people stop and ask about my TESLA (or, why charging takes zero time) – Electric Vehicle Batteries
I like the data presented here. I would suggest for every Carbon (12) forms one CO2 (44),
so 44/12 = 3.66 or 3.7 x 5.5lbs = 20.17 pounds of CO2. (instead of 19 pounds)
Lets talk about the Oxygen depletion.
For every gallon of gasoline (6.3 lbs), 13% is Hydrogen (1.2 lbs) & 87% is Carbon (5.5 lbs).
Two molecules of Hydrogen combine with one Oxygen to form water (H20 whose atomic weight is 18), so 7.2 lbs of water is created. The total Oxygen taken from the atmosphere is 6.0 lbs (for H2O) + 13.9 lbs (for CO2) for a total of 19.9 lb of Oxygen used to burn one gallon of gasoline.
All of these calculations are cool, but are leaving a huge part of the ecological puzzle out of the picture, first you are not getting “more carbon” (Energy is neither destroyed, nor created) all that carbon is exactly the same carbon, nothing of it increased! just added two atoms of Oxygen- now you have the mega fuel- “Carbon Dioxide” for the forests and flora of the earth to go into hyper drive and produce more Oxygen and wood and food, My Mother who only went to fourth grade figured this out years ago when “Smart” people were talking about the “Horrible ‘Green House gas'”, it is preposterous how we get so tunnel vision!
How about seven years late? My question: the above describes the direct emission of CO2 for a gallon of gas. But, a more useful analysis would take into account the emission of CO2 to *produce* that gallon of gas.
This is more complicated, because different extraction methods and refineries have different efficiencies, and of course in the future, we might find that those methods and refineries are even able to use renewable energy. But it seems like for now, we ought to at least be able to come up with a reasonably good estimate of the average.
Any ideas what that average is?
About 6 lb/gal for gasoline, and 5.3 lb/gal for diesel
Source: Innovation Origins in the Netherlands
This little mistake of semantics always aggravates me. It’s not 3.6 times more than 5.3. It’s 3.6 times as much. 3.6 times more than would add 3.6×5.3 to the original 5.3, which would be 24.3 pounds
Pingback: Tallying Emissions Becomes a New Challenge for Companies – All Sides Now
The main error here is that vehicles burning gasoline do not purely produce Carbon Dioxide, they produce CO, NO, HC, H2O, CO2, N2, and potentially others depending on the composition of the fuel and air. Some of those are reduced by the catalytic converter, so the whole premise is wrong. However, humans do and the current world population is ~7.978 billion with the average human generating ~1 kg (2.2lbs) of CO2 per day just by breathing. That works out to be ~8 Million metric tonnes (~8.77M US tons) per day and ~2.912 Billion metric tonnes aka GtCO2 (~3.2B US tons) per year from regular human breathing. That’s ~ 8.4% of the global total for 2021. When discussing topics like pollution it’s imperative that you consider the real daily role humans play, not a pretend role that vehicles might play. Too often variables are removed to make the math easier.